2Department of Gastroenterology, School of Medicine, Marmara University, Istanbul, Türkiye
Abstract
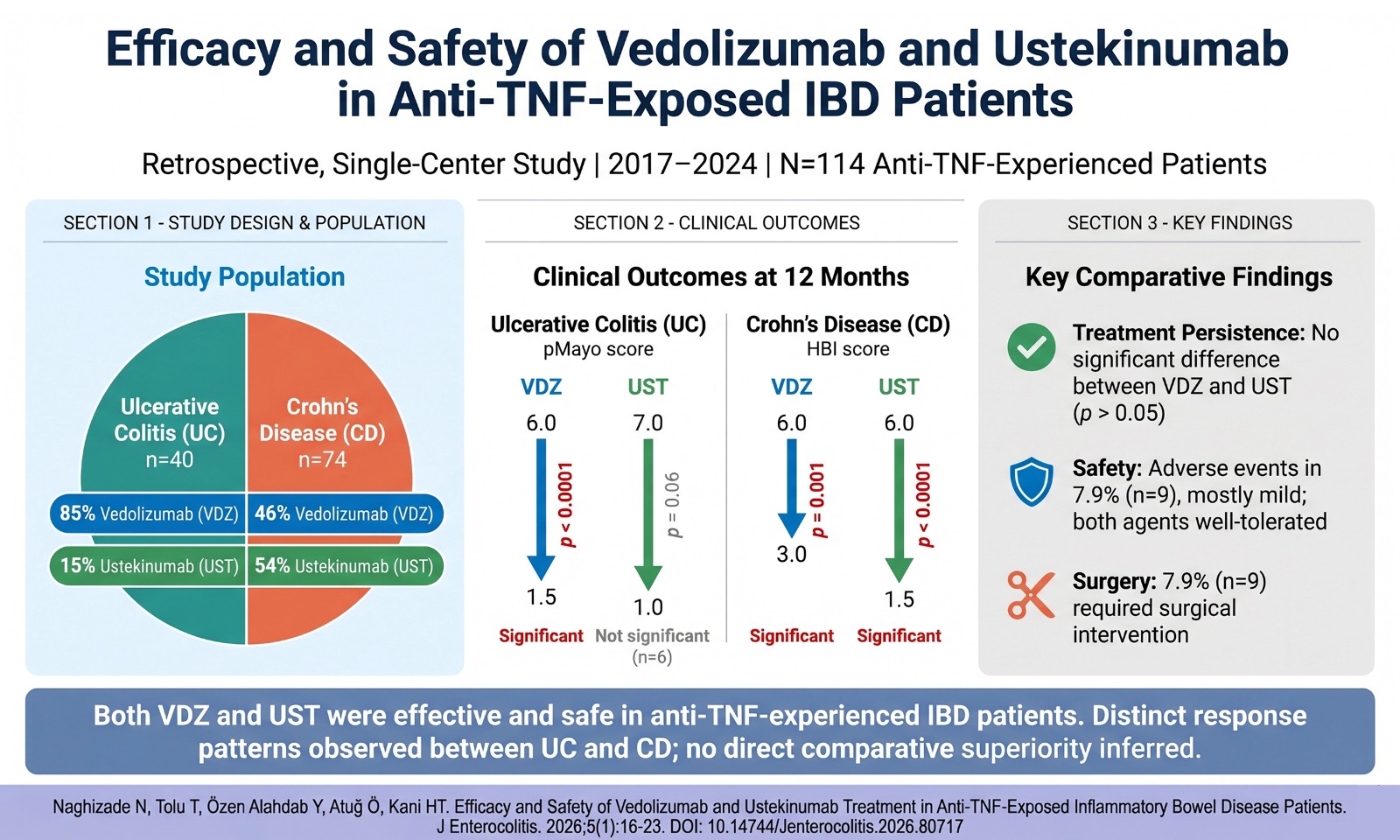
Objective: Patients with inflammatory bowel disease (IBD) who fail anti–tumor necrosis factor (anti-TNF) therapy require alternative biologics with different mechanisms of action. Vedolizumab (VDZ) and ustekinumab (UST) are established effective options. However, real-world comparative data remain limited.
Methods: We conducted a retrospective, single-center study of 114 anti-TNF-experienced IBD patients (40 ulcerative colitis [UC], 74 Crohn’s disease [CD]) treated with VDZ or UST between 2017 and 2024. Clinical and laboratory parameters were collected at baseline, at 3 and 12 months, and at the last follow-up. Disease activity was assessed using the partial Mayo score (UC) and the Harvey–Bradshaw Index (CD). Treatment response, persistence, adverse events, and the need for surgery were analyzed.
Results: Of the 40 UC patients, 34 (85%) were treated with VDZ and 6 (15%) with UST. Among the 74 CD patients, 34 (46%) were treated with VDZ and 40 (54%) with UST. In UC, VDZ led to significant reductions in pMayo scores at 3 and 12 months (p<0.0001), whereas UST showed numerical improvement without statistical significance. In CD, both VDZ and UST significantly reduced HBI scores at 3 and 12 months (p<0.001). Treatment persistence did not differ significantly between VDZ and UST in the overall, UC, or CD cohorts. Adverse events occurred in 9 patients (7.9%), mostly mild, with no serious complications. Surgical interventions were required in 9 patients, most of whom were treated with VDZ.
Conclusion: Both agents were effective and safe in anti-TNF-experienced IBD patients. Our real-world data indicate distinct response patterns between UC and CD, underscoring the clinical utility of both agents as therapeutic options after anti-TNF failure.


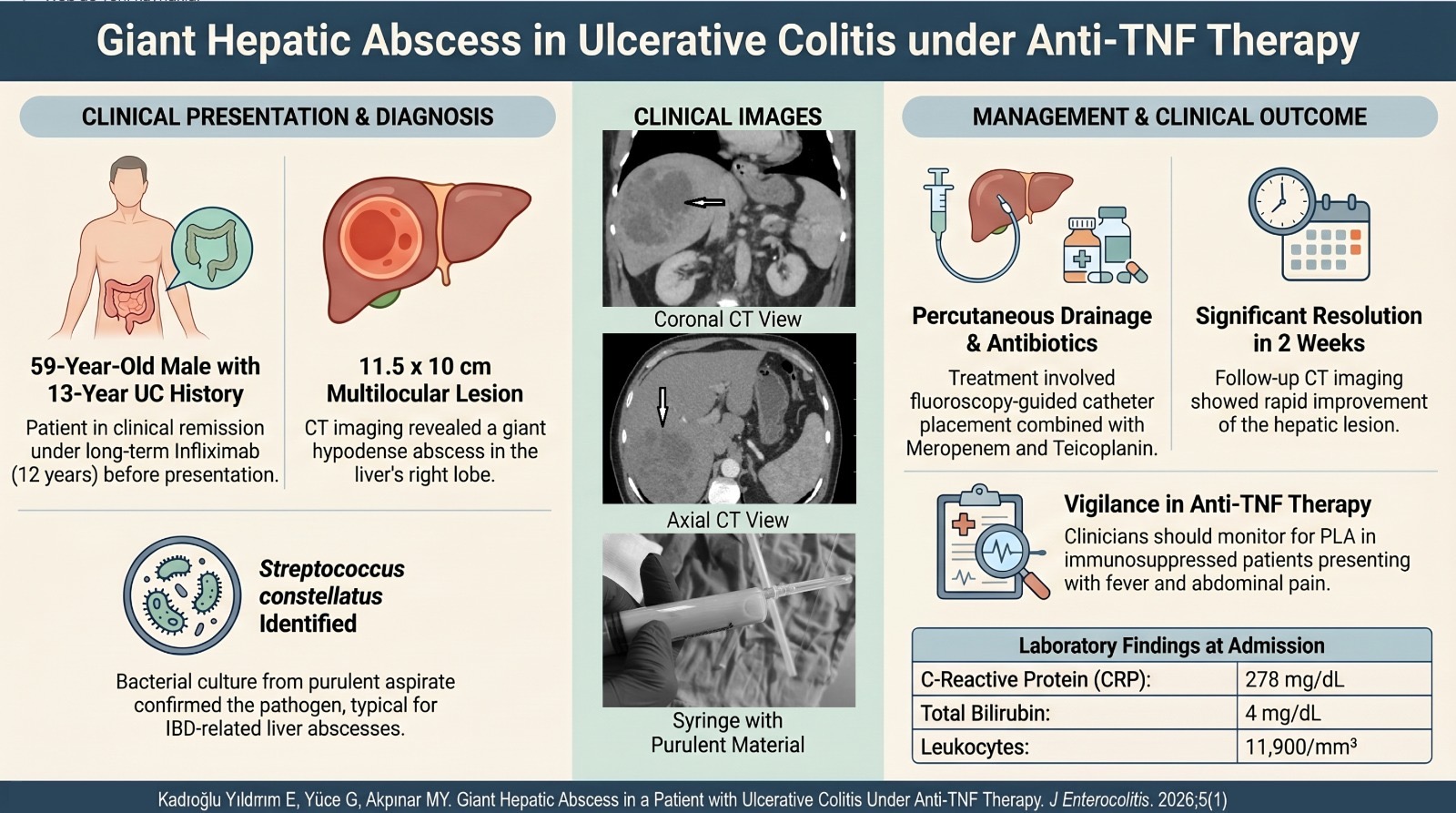


 Narmin Naghizade1
Narmin Naghizade1