2Division of Oncology, Department of Internal Medicine, Hacettepe University, Ankara, Türkiye
3Division of Gastroenterology, Department of Internal Medicine, Hacettepe University, Ankara, Türkiye
Abstract
Objective: Immune checkpoint inhibitors are an increasingly important treatment option in cancer treatment; however, potential immune-related adverse events are one of the main factors limiting the use of the treatment and colitis/diarrhea is one of them. Data about this side effect were derived from clinical trials, and articles examining this side effect in real life are limited. We evaluated the frequency and management of colitis and diarrhea in cancer cases treated with an immune checkpoint inhibitor.
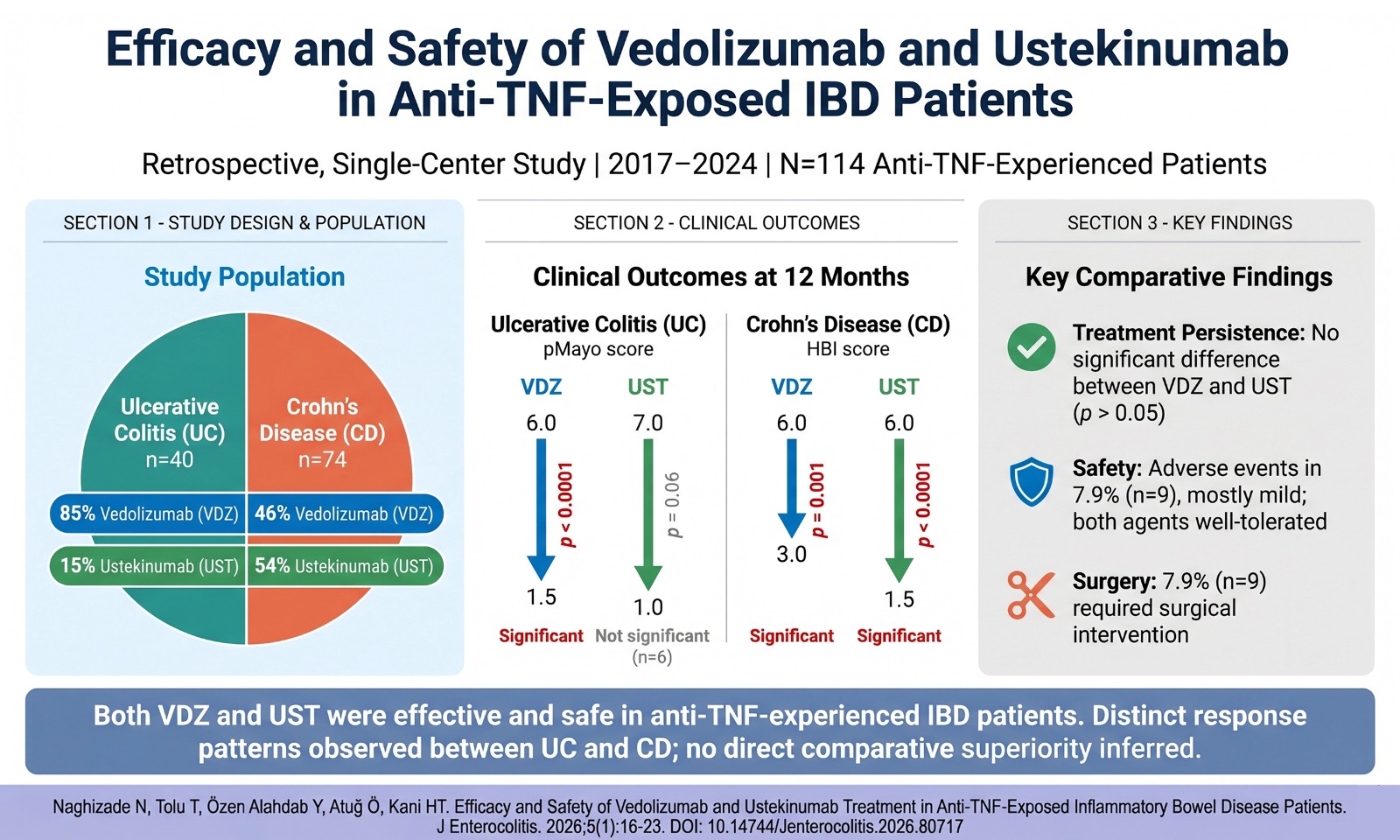
Methods: A total of 363 oncology patients treated with immune checkpoint inhibitors at Hacettepe University Oncology Hospital were evaluated retrospectively between October 2014 and November 2021. Grade 3 and higher colitis/diarrhea cases were detected in3 7 patients, and clinical properties were reviewed in detail.
Results: Of these 7 patients, 3 of them were diagnosed with renal cell carcinoma, 2 with lung adenocarcinoma, 1 with malignant melanoma, and 1 with endometrial cancer. Three patients were treated with nivolumab, 1 with pembrolizumab, 1 with atezolizumab, and 2 with nivolumab–ipilimumab combined therapy. The colitis/diarrhea occurred approximately at the end of the second month of treatment. Immunotherapy was discontinued in 6 patients. All patients were treated with steroids, and none of them needed non-steroid immunosuppressive drugs.
Conclusion: In our study, the rate of diarrhea/colitis of grade 3 and above was 2%, similar to other clinical trials. Although our patient population is small, the development of colitis in 2 patients receiving combined immune control inhibitors is remarkable. Close follow-up of patients under treatment regarding side effects protects them from possible complications with early intervention.




 Yunus Kaygusuz1
Yunus Kaygusuz1