2Department of Gastroenterology, Teaching hospital of the Ludwig-Maximilian University, Munich, Germany
3Department of Gastroenterology, Memorial Hospital, Diyarbakır, Türkiye
4Department of General Surgery, University of Health Sciences, Fatih Sultan Mehmet Training and Research Hospital, İstanbul, Türkiye
5Department of Gastroenterology, University of Medeniyet, Prof. Dr. Suleyman Yalcin Hospital, İstanbul, Türkiye
Abstract
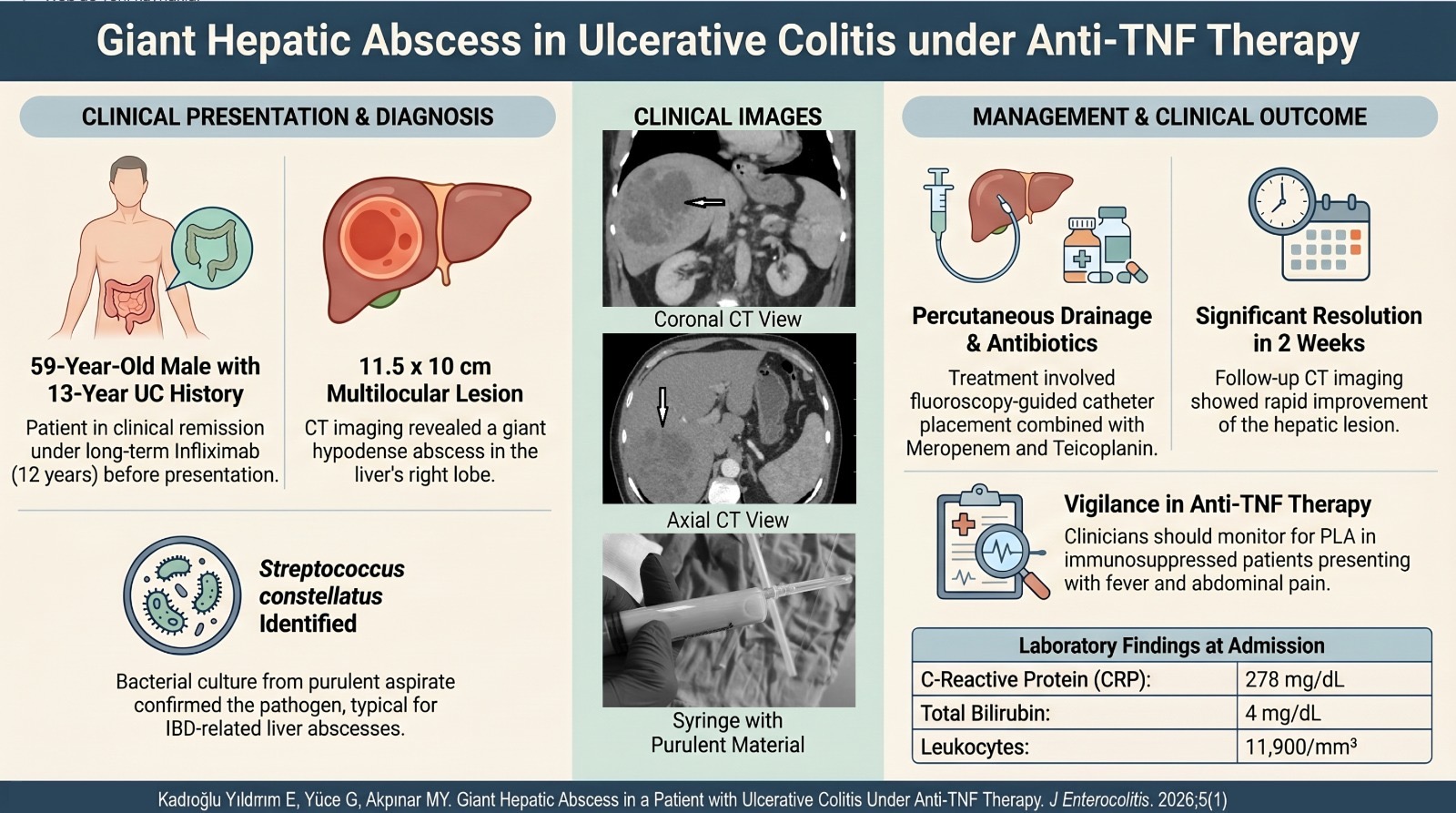
Objective: Bone mineral density is usually decreased in inflammatory bowel diseases. Patients with inflammatory bowel diseases seem to have a multifactorial pathogenesis of their osteoporosis. The aim of our study is to determine the risk factors affecting bone mineral density in inflammatory bowel diseases. The aim of our study is to determine the risk factors affecting bone mineral density in inflammatory bowel diseases.
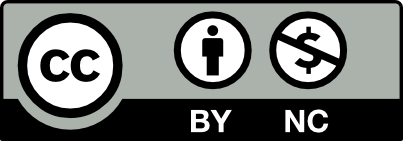
Methods: This is a cross-sectional, multicenter study involving 121 ulcerative colitis, 129 Crohn’s disease patients, and 65 healthy controls.
Results: The difference between Crohn’s disease and healthy controls in terms of osteopenia, osteoporosis, low bone mineral density, and T- and Z-scores were strongly significant (P < .01). Also the difference between T- and Z-scores in terms of osteopenia and low bone mineral density was strongly significant between ulcerative colitis and healthy controls (P < .01), in terms of osteoporosis, it was significant between ulcerative colitis and healthy controls (P < .05). In logistic forward stepwise analysis, disease activity and >5 g cumulative corticosteroid dose use were determined as risk factors for osteopenia, osteoporosis, and low bone mineral density in Crohn’s disease. Disease activity and use of >5 g corticosteroid for osteopenia, body mass index and >5 g corticosteroid use for osteoporosis, and >5 g corticosteroid use for low bone mineral density were determined as risk factors in ulcerative colitis.
Conclusion: Bone mineral density in inflammatory bowel diseases is significantly lower than that in healthy individuals. Low bone mineral density is mostly found in Crohn’s disease. Factors affecting bone mineral density are multifactorial in inflammatory bowel diseases, and it is widely accepted that bone mineral density is reduced. It is important to perform dual-energy x-ray absorptiometry regularly for patients with risk factors.
INTRODUCTION
Inflammatory bowel diseases (IBD) are divided into Crohn's disease (CD) and ulcerative colitis (UC). Bone mineral density (BMD) is usually decreased in IBD.1 The factors leading to osteoporosis (OP) in IBD are multifactorial and can be divided into 2 groups as non-modifiable (e.g., genetic factors, age, female gender, menopause) and modifiable (e.g., sedentary lifestyle, alcohol, smoking, corticosteroids (Cs), hypogonadism, calcium and vitamin D deficiency, malnutrition, low body mass index (BMI)).2-4 There are opinions that interleukin (IL)-6 may cause loss of BMD by affecting osteoblast differentiation whereas NOD2 mutations do not pose a risk for loss of BMD.5,6 However, patients with newly diagnosed CD who have not received prior Cs have low BMD.6 Combination therapy with an immunomodulator + anti-TNF alpha (tumor necrosis factor) increases BMD in IBD patients. This is the rationale for early aggressive anti-inflammatory therapy of IBD patients with a complicated disease course and disease activity.7 Age-related bone loss may be slowed down by vitamin D and calcium supplementation.8 In IBD patients, negative systemic calcium balance is considered as one of the important factors in association with vitamin D deficiency.9
METHODS
Patients and Study Design
This is a cross-sectional, multicenter study involving 121 UC, 129 CD patients, and 65 healthy controls (HC) including patients from 3 hospitals. It was conducted between January 2018 and July 2021. The diagnosis of UC and CD was made according to clinical, radiographic, endoscopic, and histopathological features. Demographic and clinical features of all patients, including age, gender, age at diagnosis, duration and localization of disease, menopausal status, smoking, and used drugs were recorded. All data were analyzed anonymously. The BMDs of the lumbar spine (L2-L4), left femoral neck, and total femur were measured by dual-energy x-ray absorptiometry (DXA; Prodigy, GE Medical systems Lunar, Wis, USA) reported as BMD T- and Z- scores. Low BMD was defined according to World Health Organization criteria. T-scores greater than 1 standard deviation (SD) were categorized as normal, while T-scores between 1.0 and 2.5 SD and less than 2.5 SD were classified as diagnostic for osteopenia (OPe) and OP, respectively.10
Patients with diabetes mellitus, cancer, pregnancy, diseases that alter bone mass (such as thyroid disease, chronic kidney failure, chronic obstructive pulmonary disease, chronic liver disease, and systemic lupus erythematosus) and postmenopausal women under hormonal replacement therapy were excluded. The same exclusion criteria were used in the control group, with no difference in age and gender according to UC and CD. Healthy controls consisted of healthy adults. The control group included humans without the use of drugs affecting bone turnover and who had no metabolic bone disease or bowel resection previously.
Statistical Analysis
The statistical analysis was carried out using Statistical Package for the Social Sciences version 26.0. (IBM SPSS Corp.; Armonk, NY, USA). The Shapiro–Wilk test was used as a homogeneity test. Mann–Whitney U test of significance when appropriate for quantitative data as significance test, chi-square test for qualitative data, and logistic stepwise forward regression analysis were used to identify risk factors. Unless otherwise stated, results are given as mean ± SD. To compare the distribution of normal BMD, OPe, and OP between the 2 patient groups, 2 × 3 contingency tables were used for the chi-square test. P < .05 was accepted as significant and P < .01 was accepted as strongly significant.
RESULTS
A total of 315 subjects, 129 with CD (71 male), 121 with UC (65 male), and 65 HC (33 male) were admitted to the study. The mean ages of CD, UC, and HC were 39.9 ± 13.5, 41.14 ± 12.6, and 42.38 ± 12.7, respectively. There was no difference between the groups regarding age and gender (P > .05) (Table 1).
While the difference in BMD between CD and UC was significant (P < .05), the difference was strongly significant in relation to T- and Z- scores (P < .01). The difference between CD and HC in terms of OPe, OP, low BMD, T- and Z- scores was strongly significant (P < .01), while the difference between T- and Z- scores in terms of OPe and low BMD was strongly significant between UC and HC (P < .01) and in terms of OP was significant between UC and HC (P < .05) (Table 2).
In CD, disease duration affected OPe, OP, and low BMD (P = .032, P = .018, P = .013, respectively). Azathioprine affected OP (P = .049). Disease activity was significant with OPe (P = .001) and strongly significant with OP and low BMD (P < .001). The relationship between Cs>5 g and OPe, OP, and low BMD was strongly significant (P < .001) (Table 3).
In UC elderly patients, late onset of disease and low BMI affected OP (P = .036, P = .023, P = .003 respectively). Both disease activity and Cs>5 g were strongly significant (P < .001) with OPe, OP, and low BMD (Table 4).
In CD, logistic forward stepwise analysis revealed that disease activity and >5 g Cs usage were risk factors for OPe, OP, and low BMD. The results of the risk analysis for disease activity regarding OPe, OP, and low BMD in CD were as follows: odds ratio (OR): 7.295 (95% CI: 1.473-36.138), P = .015; OR: 9.369 (95% CI: 1.678-52.307), P = .011; and OR: 7.47 (95% CI: 1.601-34.859), P = .11, respectively. When risk analysis was performed in CD patients taking Cs, we had the following results regarding OPe, OP, and low BMD: OR: 11.624 (95% CI: 2,439-55.391), P = .002; OR: 7.32 (95% CI:1.273-42.09), P = .026; OR: 10.091 (CI 95%: 2.199-46.302), P = .003 respectively (Table 5).
In UC, disease activity and use of >5 g Cs were defined as risk factors for OPe whereas BMI and >5 g Cs usage were defined as risk factors for OP, and for low BMD, >5 g Cs usage was defined as a risk factor in the logistic forward stepwise analysis. The correlation with BMD in UC was again inverse, with active disease being significantly associated with OPe, OP, and low BMD. A correlation was found with OPe in risk analysis in UC (OR: 3.462 (95% CI: 1.018-11.777), P = .047). The risk analysis for UC patients taking Cs regarding OPe, OP, and low BMD was as follows: OR: 4.268 (95% CI: 1.284-14.19), P = .018; OR: 11.327 (95% CI: 1.771-72.46), P = .01; OR: 5.239 (95% CI: 1.67-16.43), P = .005, respectively (Table 6).
DISCUSSION
The mechanism of bone loss is not definitely answered yet. Bone loss initially was being attributed to the use of drugs such as Cs. However, low BMD is observed in studies conducted with IBD patients without any treatment, implying that this inflammatory mechanism contributes to the process.11 Disease duration and activity are associated with lower BMD values.12 Some drugs used to treat IBD cause changes in BMD. It is well known that Cs use is a risk factor for OP, whereas AZA and anti-TNF-alpha therapy are known to contribute to increasing bone mass.4,13 A decrease in BMD is related to a raised risk of fracture. Patients with IBD have a 40% higher risk of fractures than the normal population, resulting in increased morbidity and reduced quality of life.13 Bone fractures were not noted in our study, so we were unable to provide a reliable explanation and interpretation. Inadequate diagnosis and treatment of bone disease in IBD contribute to morbidity. Publications on this topic show that more than 80% of fractures in patients with IBD did not receive prior appropriate treatment for the diagnosis of OPe or OP.14 The aim of our study is to determine the risk factors affecting BMD in IBD.
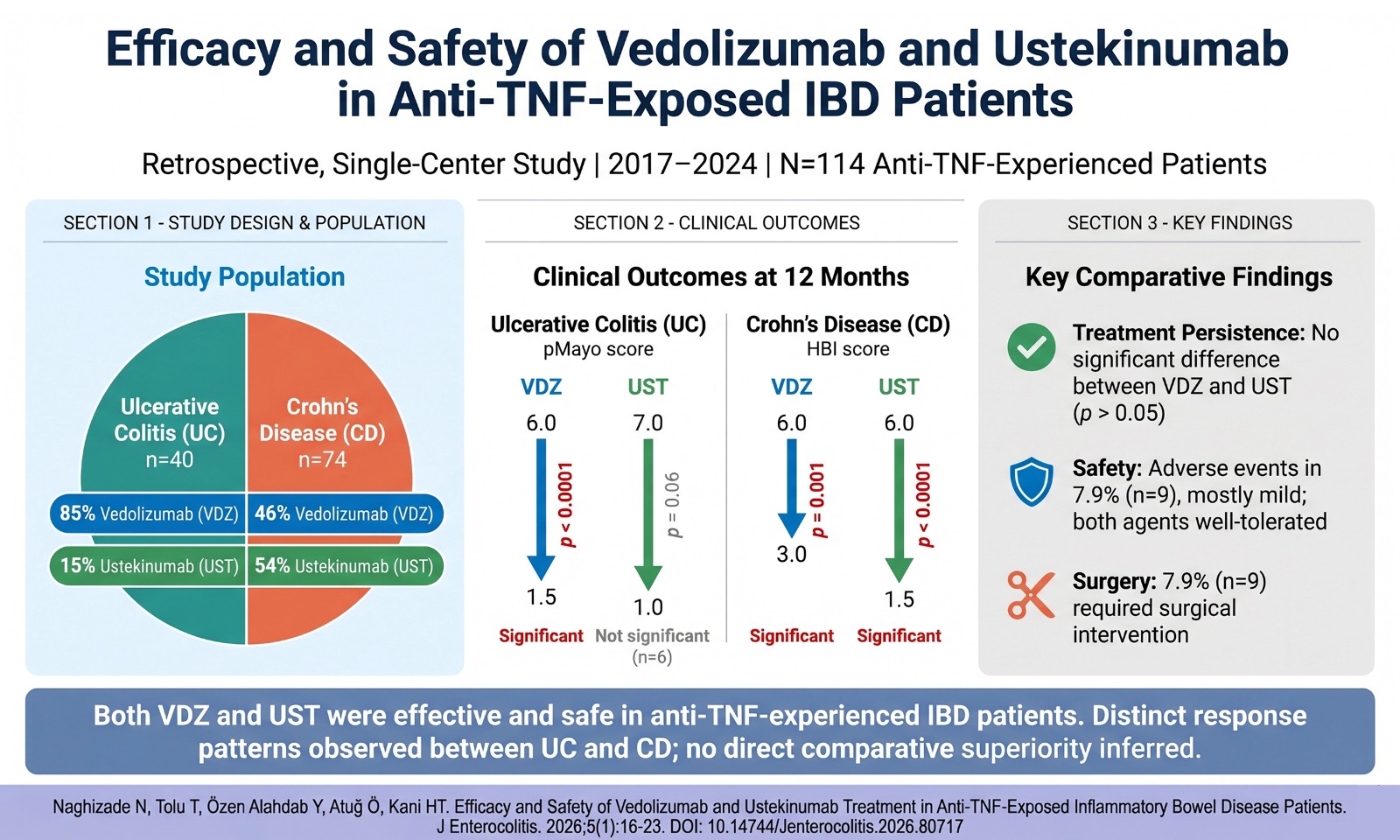
Some studies have shown a higher prevalence of low BMD in patients with CD than in patients with UC,4 while this difference is not clear in other studies. Two-thirds of patients with IBD have lower BMD than healthy individuals.15 In a study by Lima et al.16 195 subjects (60 CD, 68 UC, and 67 HC) were investigated. The majority of patients with UC (97.1%) and CD (73.3%) were in clinical remission; 28 (41.2%) of UC and 32 (53.3%) of CD patients had abnormal BMDs in the study. Osteoporosis was found in 2 (2.9%) patients in UC and in 7 (11.7%) CD patients. Low BMD was found in 3 (4.5%) subjects in the control group.
In contrast, two-thirds of the IBD patients in our study group had low BMD, 96 (74.4%) subjects in CD and 70 (57.8%) subjects in UC. In Crohn’s Disease, OPe, OP, low BMD, T-and Z-Scores were significantly lower in comparison to both UC and HC-group. Further, these values in UC were significantly lower compared with HC. The lower BMD in CD suggests that disease-related factors are more dominant. In our study, our OPe, OP, and low BMD rates were higher compared to Lima et al’s study. We think that the number of disease-active patients (44 (34.1%) in CD, 30 (24.8%) in UC) is higher and the age difference between the series is influential.
A recent study by Dar et al5 stated that IBD patients with lower BMI and use of Cs had lower BMD values, while smoking and male gender were determined as risk factors of borderline importance. In our study, IBD patients with OP had low BMI and Cs use as risk factors. Interestingly, smoking and male gender were not linked with low BMD in our study cohort. An American cohort did not find any statistically significant difference in BMD between non-smokers and smokers.17 This result was consistent with our findings.
Inflammatory bowel disease-related bone loss appears to be related to disease activity.18 In IBD, high levels of inflammatory cytokines (IL-1, IL-6, IL-11, IL-17, transforming growth factor (TGF)-α, TNF-α, epidermal growth factor, and prostaglandin E2) are determined in the intestinal mucosa and peripheral blood.19,20 The main cytokine seems to be IL-6, which correlates negatively with BMD.19 Receptor activator of NF-κB ligand (RANKL) is also induced in these patients. Plasma osteoprotegerin levels were found to be 1.9 times higher in UC and 2.4 times higher in CD, possibly representing a counterattempt to increase RANKL.20 Reffitt et al21 studied 137 subjects with IBD. Scores showed correlation between remission time and Z-score in both UC and CD. In active disease, femoral neck and lumbar spine Z-score were lower than those being in remission.
In our study, CD patients had a negative correlation between disease activity and BMD. We found a significant relationship between active disease and OPe, OP, and low BMD, which could be confirmed by a significant increase in the risk analysis for OPe, OP, and low BMD.
The correlation with BMD in UC was again inverse, with active disease being significantly associated with OPe, OP, and low BMD. The risk analysis in UC patients only delivered a significant relationship to OPe. The risk analysis in CD patients taking Cs demonstrated a 7.2, 9.3, and 7.4 times increase in OPe, OP, and low BMD, respectively. The risk analysis in UC patients with Cs usage showed a 3.4 increase in OPe.
Corticosteroid therapy is supposed as a risk factor for bone loss.12 Use of Cs leads to an imbalance between osteoblasts and osteoclasts on behalf of a shift of the osteoclasts. Corticosteroids increase osteoclast differentiation and activation leading to a decline in bone formation by inducing apoptosis of osteoblasts.22,23 Corticosteroids reduce BMD by several mechanisms, including stimulating osteoblast apoptosis, diminishing intestinal absorption of calcium, and increasing renal calcium excretion.24 Long-term Cs use reduces BMD and has been involved with reduced bone density in patients with IBD, although some studies did not find such a connection.25 In a study of 166 patients with IBD, Abraham et al26 showed that Cs use more than doubled the risk of low BMD (OR = 2.4 (1.5-3.6), P = .001). However, other studies did not reveal a clear association between low BMD and Cs use. In this sense, Andreassen et al27 found no context between BMD and Cs in a study comprising 113 CD patients. In a study by Jahnsen et al.,28 IBD patients were followed up with DXA for 2 years. No context was found between Cs use and BMD. There was no difference between the DXA measured at the end of 2 years and the initial DXA. Miznerova et al29 examined 76 IBD patients and did not found any correlation between Cs use and BMD, too. However, in another study, Silvennoinen et al30 examined risk factors and the prevalence of low BMD in patients with IBD. They showed that patients with a lifetime Cs >10 g had a significantly lower BMD compared to the Cs-free or <5 g Cs groups. In addition, there was no decrease in BMD in patients who did not receive Cs. The authors found a negative association between Cs and BMD.
According to the findings of our study, the relation between CS use 5 g and OPe, OP, and low BMD in CD and UC was strongly significant compared to patients who did not receive Cs or received CS <5 g. The risk analysis in CD patients taking Cs demonstrated an 11.6, 7.3, 10 times increase of OPe, OP, and low BMD, respectively. The risk analysis in UC patients with Cs usage showed a 4.2, 11.3, and 5.2 times increase in OPe, OP, and low BMD, respectively.
In a study by Walldorf et al.31 DXA of 173 patients with IBD was evaluated. In the forward stepwise regression analysis (weight, BMI, gender, nicotine abuse, disease pattern and duration, treatment with Cs, AZA, anti-TNF-α treatment), gender, disease duration, and Cs treatment were influential on the presence of OP. It was predictive in 80.7% of OP cases. In male patients, the OR for OP was 3.9 (P = .025). The OR increased by 1.1 (P = .012) in disease duration and 1.02 (P = .003) in CS treatment. Wada et al32 evaluated 388 subjects with IBD by DXA. Multivariate regression analysis was performed. Risk factors for UC were low BMI, Cs dose, and disease localization. Risk factors for CD were male gender and low BMI.
In our study, disease duration and AZA use are additional factors affecting BMD in CD. There was a negative context between disease duration and BMD in CD. The longer the disease duration, the more the decrease in BMD. This suggested a greater effect of the disease on BMD rather than AZA. Increasing age, late onset of disease, and low BMI negatively affected BMD, whereas low BMI was a risk factor for OP in UC, furthermore. This emphasized the importance of BMI.
The hypothesis that disease activity is an independent risk factor for BMD is strengthened by the finding that patients using Cs and AZA are important predictors of BMD. A reversal relationship of AZA with BMD has not been demonstrated. In accordance, it was hypothesized that AZA is reversely associated with BMD because of its application in IBD patients with high inflammatory activity who require additional immune-suppressive medication.11 Little is known about the effects of AZA on BMD, although 1 study reported a correlation between AZA and BMD. This could be because of the underlying drug’s property to suppress disease activity and maintain remission induction rather than its direct effect on BMD.21
In our study, there was a reversal correlation between AZA and BMD. This may be because the number of active patients is high and the remission period is not taken into account. In addition, the duration of use of AZA was not taken into account. Since long-term use of AZA will prolong the remission period, it seems more likely to increase BMD.
The strongest feature of our study is that it was a multicenter study with the patients being chosen randomly and sequentially. One of the limitations of our study was the small number of patients, second that the vitamin D level was not included in the study, and third that eventually fractures were not considered. Prospective, long-term controlled, double-blind studies with more patients are needed to obtain more reliable results.
In conclusion, BMD reduction is evident in IBD. This is multifactorial and well known. Decrease in BMD is most common in CD. Factors leading to BMD reduction in CD are AZA, disease duration, disease activity, and Cs use, whereas increasing age, late-onset, low BMI, disease activity, and Cs use affect BMD in patients with UC. In cases of patients with risk factors and reduced BMD, the beginning of appropriate treatment could reduce morbidity and mortality. For this reason, DXA should be performed in patients with risk factors and it is vital to initiate appropriate treatment for patients with low BMD. In general, we recommend to conduct DXA in IBD patients with increasing age, low BMI, long disease duration, late-onset disease, disease activity, and Cs use.




 Mehmet Koroğlu1
Mehmet Koroğlu1