2Department of Biotechnology, DiaGen Biotechnology, Ankara, Türkiye
3Department of Gastroenterology, Lokman Hekim University, Faculty of Medicine, Ankara, Türkiye
Abstract
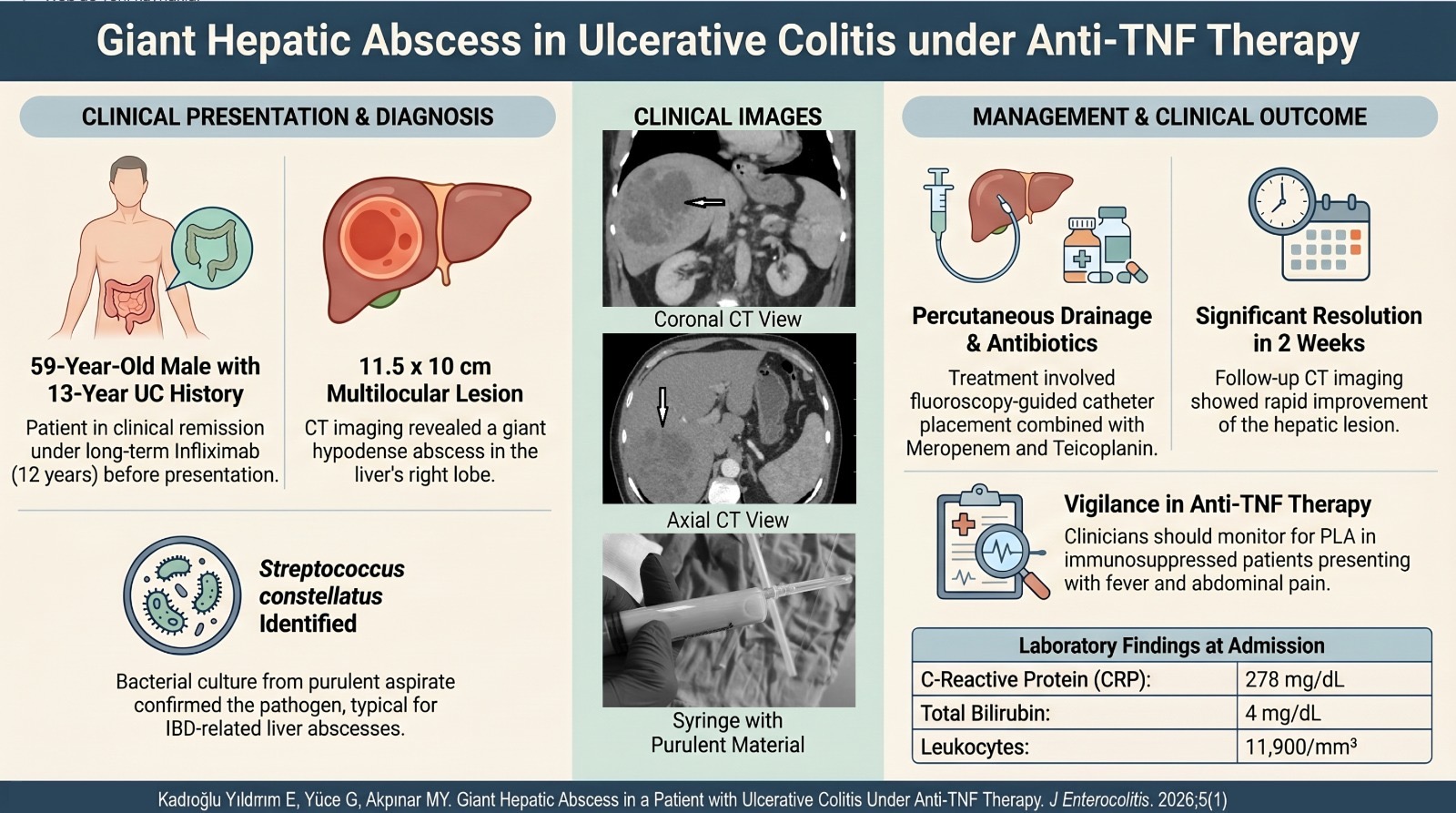
Objective: Irritable bowel syndrome is a common functional gastrointestinal disorder that affects the quality of life and the ability to work. It is known that irritable bowel syndrome occurs as a result of a multifactorial change in microbiota and immunology. We aimed to evaluate the effects of Bacillus subtilis on inflammatory parameters and quality of life in irritable bowel syndrome.
Methods: Irritable bowel syndrome volunteers diagnosed according to Rome IV criteria were recruited in the single-center prospective study and were given 5 billion B. subtilis once a day for 3 weeks. The primary outcome, the irritable bowel syndrome symptom severity score, fecal microbiota analysis, and serum inflammatory parameters were evaluated at the beginning of the study and at the end of the 3-week study period.
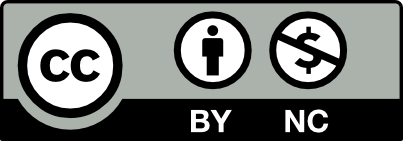
Results: The mean age of 22 patients included in the study was 39.90 ± 11.93 years. The mean irritable bowel syndrome symptom severity score was 144.31 ± 104.86 before the treatment and 62.50 ± 43.69 after the treatment and the difference between them was statistically significant (P < .001). After treatment, an increase in total oxidant status, total antioxidant status, oxidative stress index, interleukin-10, interferon-gamma levels, and a decrease in interleukin-6 and tumor necrosis factor-α levels were observed, but the difference was not statistically significant (P > .05). While there was no difference in Shannon diversity after the application of alpha diversity analysis, a decrease was observed in the Chao (P < .006) analysis.
Conclusion: The use of probiotics containing B. subtilis spores in patients with diarrheal-type irritable bowel syndrome reduces the severity of the disease, but no significant effect was observed in terms of inflammatory parameters.
INTRODUCTION
Irritable bowel syndrome (IBS) is a common, chronic gastrointestinal condition defined by alterations in bowel habits and abdominal pain, in the absence of organic pathology. One-third of patients admitted to the gastroenterology clinics have functional gastrointestinal disorders, and the most common diagnosis is IBS.1 Irritable bowel syndrome treatment includes patient education, diet and lifestyle changes, and medical treatment. It has been suggested that changes in the gut microbiota cause IBS.2 Studies comparing the gut microbiota of IBS patients with healthy controls have shown an altered microbiota profile in IBS, and specific gut microbiota profiles have been associated with certain symptoms and severity of the disease.3
A rod-shaped, spore-forming, Gram-positive facultative aerobic bacterium, Bacillus subtilis, has recently been considered for its probiotic poten-tial. In addition, recent evidence indicates that B. subtilis is part of the normal gut microbiota of humans and may play a critical role in immune development and supporting digestive health.4 In this study, we aimed to investigate the effects of B. subtilis on patients’ quality of life and inflam-matory parameters in IBS.
METHODS
Study Population
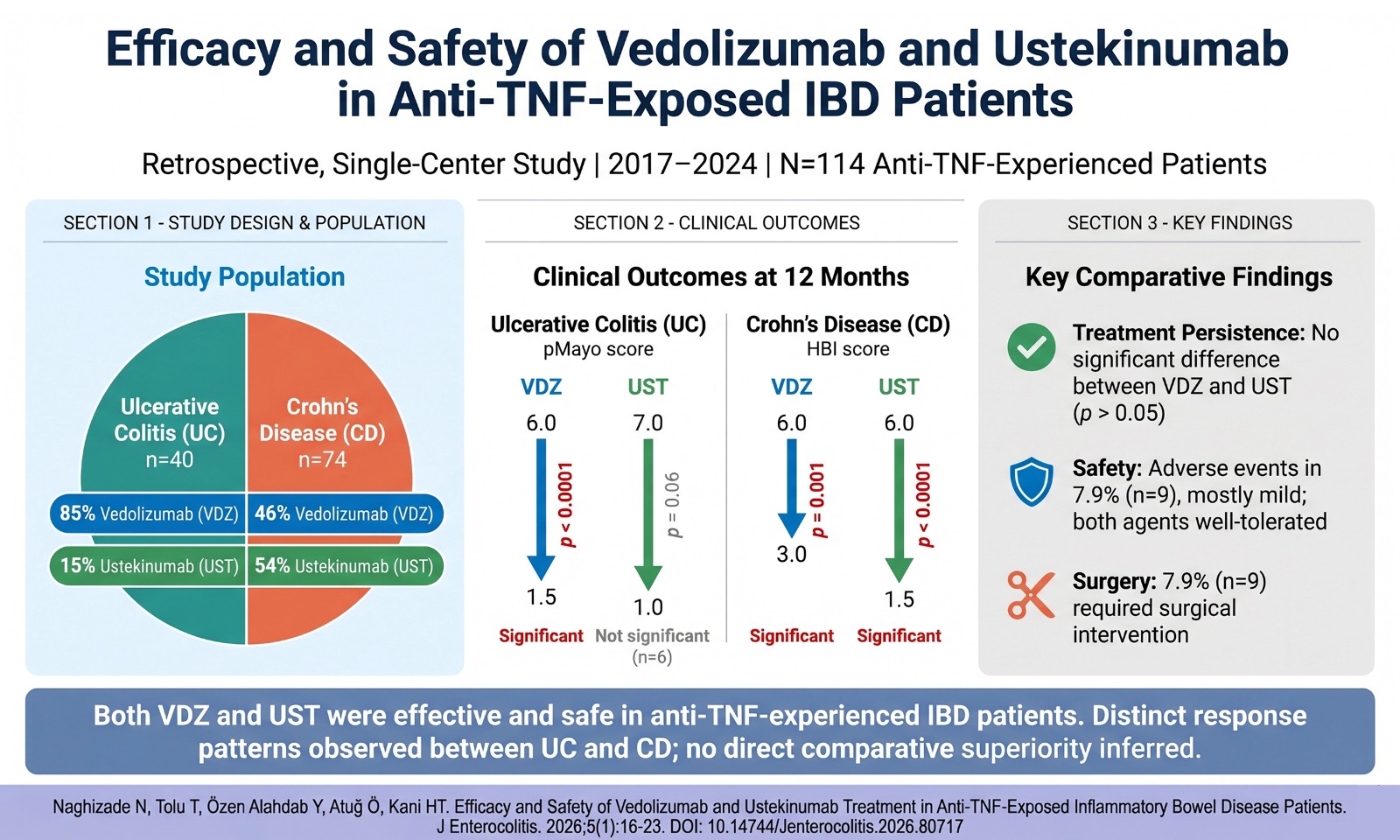
This study was planned as a prospective single-center study. Patients aged 18-65 years, admitted to the gastroenterology outpatient clinic between July 2021 and October 2021, diagnosed with IBS according to the ROMA IV criteria, and classified as diarrheal-type IBS according to the ROMA IV classification were included in the study. Ethics committee approval was received from Lokman Hekim University for this study (Approval number: 2021/54). All participants confirmed their informed consent.
Inclusion and Exclusion Criteria
Patients between the ages of 18 and 65 whose underlying organic disease was excluded by gastroscopy and colonoscopy were included in the study.
Patients with alarm symptoms such as unintentional weight loss, dysphagia, unexplained iron deficiency anemia, persistent vomiting, family history of upper gastrointestinal cancer, patients with active infection and recent infection, those with gastroscopically or colono-scopically proven pathological findings, psychiatric drug use, pregnant and lactating patients, patients who used probiotics and antibiotics in the last 4 weeks, morbid obesity, any chronic autoimmune disease, or malignancy were excluded from the study. Inflammation plays an important role in the development and maintenance of diabetes and hypertension. We also excluded patients with diabetes and hyperten-sion because we looked for inflammatory markers in these patients.
Patients were given 1 vial (10 mL) of deionized water containing 5 billion B. subtilis spores once a day for 3 weeks. The patients took the preparation in the morning on an empty stomach, 1 vial was added to 1 glass of water, held for 5 minutes, and then was taken. Irritable bowel syndrome symptom severity score (IBS-SSS), leukocyte, eryth-rocyte sedimentation rate (ESR), c-reactive protein (CRP), neutr ophil -lymp hocyt e ratio (NLR), platelet/lymphocyte ratio (PLR), mean plate-let volume (MPV), total oxidant status (TOS), total antioxidant status (TAS), oxidative stress index (OSI), tumor necrosis factor (TNF)-alpha, interleukin (IL)-10, interferon (IFN)-gamma, IL-6 levels were measured before and after treatment. Fecal microbiota analysis was performed on patients before and after treatment.
EVALUATED PARAMETERS
Disease Severity
Disease severity in patients was evaluated 1 week before the start of treatment and within 1 week after the end of the treatment. To deter-mine the IBS-SSS, Francis et al.’s scoring system that measures the severity of abdominal pain, the number of days with abdominal pain, bloating, bowel habits, and the effect of each on quality of life was used. In this scoring, each question is given a score between 0 (not impaired) and 100 (highly impaired), and the total score of the ques-tionnaire can be 0 to 500, with the highest IBS-SSS score of 500. Mild, moderate, and severe cases are indicated by scores of 75-175, 175-300, and over 300, respectively.5
Inflammation Markers
Parameters such as leukocytes, ESR, CRP, NLR, PLR, and MPV were evaluated with routine blood tests.
Blood samples taken from patients before and after treatment to evaluate markers such as TNF-alpha, IL-10, IFN-gamma, and IL-6 were taken and centrifuged at low rpm (3000 rpm, G Force = 1107); separated serums were taken into 1.5 mL Eppendorf tubes and kept at −80 degrees until the date of analysis. Analyses were performed with the Biotek TS 800 ELISA reader.
Studies have shown that the production of reactive oxygen species associated with both oxidative stress and inflammation may play a role in the pathogenesis of IBS. In order to determine the general oxidant and antioxidant status in the body, markers such as TOS and TAS have been developed. The OSI is also used as a general indicator of oxida-tive stress.6
Total antioxidant status levels were measured using commercially avail-able kits (Relassay, Gaziantep, Turkey). The novel automated method is based on the bleaching of characteristic color of a more stable 2,2′- azino -bis( 3-eth ylben zothi azoli ne-6- sulfo nic acid) radical cation by antioxidants. The assay has excellent precision values, which are lower than 3%. The results were expressed as mmol Trolox equivalent/L.
Total oxidant status levels were measured using commercially avail-able kits (Relassay). In the new method, oxidants present in the sample oxidized the ferrous ion-o-dianisidine complex to ferric ion. The oxida-tion reaction was enhanced by glycerol molecules abundantly present in the reaction medium. The ferric ion produced a colored complex with xylenol orange in an acidic medium. The color intensity, which could be measured spect ropho tomet rical ly, was related to the total amount of oxidant molecules present in the sample. The assay was calibrated with hydrogen peroxide, and the results were expressed in terms of micromolar hydrogen peroxide equivalent per liter (μmol H2O2 equivalent/L).
The ratio of TOS to TAS was accepted as the OSI. For calculation, the resulting unit of TAS was converted to μmol/L, and the OSI value was calculated according to the following formula: OSI (arbi-trary unit) = TOS (μmol H2O2 equivalent/L)/TAC (μmol Trolox equivalent/L).
FECAL MICROBIOTA ANALYSIS
DNA Extraction
For the extraction protocol, the QuickGene (DNA extraction kit from tissue) extraction device was used. First, 25 mg of stool sample was transferred to the homogenization tube with 250 µL of MDT (Tissue Lysis) solution. For homogenization, 15 mg of 0.1 mmø glass beads or 10 pieces of 1.0 mm zirconia beads were added to the tube. Homogenization was performed for 2 × 120 seconds at 5000 rpm in the homogenizer. After the sample was homogenized, 25 µL of EDT (Proteinase K) solution was added and incubated at 56˚C for 60 min-utes. Then, it was centrifuged at 15 000 g for 10 minutes at room tem-perature. After centrifugation, 200 µL of supernatant was transferred to a 1.5 mL microtube. After 180 µL of LDT (Cell Lysis) solution was added and vortexed for 15 seconds, the microtube was left to incubate at 70˚C for 10 minutes. In the next step, 240 µL of 99% cold ethanol was added and vortexed for 15 seconds. The entire contents of the microtube were transferred to the QuickGene (Kurabo) filtered cassette and washes and elutions were performed following the instrument protocol. Three washes were performed using 750 µL of WDT (wash buffer) solution. As a result of the extraction process, an average of 50-60 ng of genomic DNA diluted with 200 µL of CDT (elution buffer) was obtained.
Polymerase Chain Reaction and Sequencing
The 16S rDNA V3-V4 region in the extracted bacterial DNAs was amplified by polymerase chain reaction (PCR) using 341F CCTAYGGGRBGCASCAG 806R GGACT ACNNN GGGTA TCTAA T primers, and sequencing was performed following the 2 × 250 bp pair-end protocol on the HiSeq platform (Illumina, San Diego, USA) where approximately the entire region overlapped. These primers used during amplification include both adapters for HiSeq sequencing and single-end barcodes for direct sequencing of PCR products and creating an on-chip pool system.
Bioinformatics Analysis
Pair-end Illumina reads (150 bp) were transferred to qiime2 (2019.10). Quality trimming of reads, chimera detection, and cleaning was done through the qiime2-Dada2 pipeline. Amplicon sequence variants gen-erated by Dada2 were mapped to the GreenGenes (http: //gre engen es.lb l.gov ) database. Phyloseq1 objects were created from qiime2 structures in the R4.1 environment. Alpha diversity assessment, which was used to evaluate the diversity of related taxonomic units in a sample, was interpreted using 3 different indices: Chao1, Shannon, and Simpson. The Kruskal–Wallis statistical test was used to show the distribu-tion differences between the groups. Beta diversity analysis, used to evaluate taxonomic differences between groups, was calculated based on weighted and unweighted unifrac. Specific taxonomic differences between groups were determined by differential abundance analysis, DEseq23 (R package).
RESULTS
The mean age of 22 patients included in the study was 39.90 ± 11.93 years. The mean IBS-SSS score of the patients was 144.31 ± 104.86 before the treatment and 62.50 ± 43.69 after the treatment, and the dif-ference between them was statistically significant (P < .001). A statisti-cally significant decrease was detected only in leukocyte values after treatment (P = .007) when comparing the hemogram parameters, CRP, ESR, before and after treatment. A decrease was observed in Hb, CRP, and MPV after treatment, but it was not statistically significant (P > .05). A statistically insignificant increase was observed in NLR and PLR ratios after treatment (P > .05). After treatment, an increase in TAS, TOS, OSI, IL-10, IFN-gamma levels, and a decrease in IL-6 and TNF-α levels were observed, but the difference was not statistically significant (P > .05). The analysis results of all parameters evaluated in the study group are summarized in Table 1.
While there was no difference in Shannon diversity after the applica-tion of alpha diversity analysis, a decrease was observed in the Chao diversity index (P < .006). This shows that the post-treatment group is similar to each other (Figure 1).
In the principal component analysis graph, which is a beta diversity analysis, it is seen that there was a narrowing in the group after the treatment, and the samples in the group showed similar characteristics.
In the current study, the predominant members of the microbiota were Bacteroides caccae, Prevotella stercorea, Ruminococcus flave-faciens, and Dorea formicigerans before treatment. After probiotic treatment, dominant microbes were Butyrivibrio crossotus (P = .042), Ruminococcus callidus (P = .014), and Bifidobacterium bifidum (P = .021) (Figure 2).
DISCUSSION
Probiotics are used in cases of postinfectious IBS and antib iotic- associate d diarrhea.7 Although there are studies on their efficacy in IBS, current guidelines do not recommend the routine use of probiot-ics in IBS.8 Probiotics are thought to modulate the host immune system apart from stabilizing the gut microbiota and preventing gastrointesti-nal disorders. Some studies show that the immunomodulatory effects of probiotic may be strain dependent.9 In a study by Horosheva et al10, B. subtilis strains were shown to reduce antib iotic -asso ciate d diarrhea and antibiotic side effects. In a study by Freedman et al.9 it was shown that B. subtilis can cause an increase in the anti-inflammatory immune cell population.
B. subtilis is widely used to produce fermented foods in eastern Asia. It has been considered for its probiotic potential due to its ability to form resistant spores that increase their survival in the gastrointestinal tract. Studies have shown that B. subtilis is part of the normal gut microbiota of humans and can play a critical role in immune development and supporting gastrointestinal system health.4,11,12 The exopolysaccharide produced by B. subtilis to create an aerobic microenvironment in the gastrointestinal tract can also act as a prebiotic.13
Biological diversity and richness are critical to the maintenance of normal physiological functions of the host. Studies have shown that B. subtilis supplementation increases biodiversity and stimu-lates the immune system.14-16 In this study, there was no significant increase between the pre- and post-treatment groups in the analysis of alpha and beta diversity. Even alpha diversity analysis showed a decrease in the Chao index. However, when we compared the inflammatory parameters, there was only a significant decrease in leukocyte values. There was a non-significant decrease in CRP and MPV after treatment.
In this study, it was observed that B. crossotus, R. callidus, and B. bifi-dum were dominant after probiotic treatment. It has been observed that there is a correlation between B. crossotus and xylanase/xylosidase.17 They are also producers of butyrate, lactate, and acetate. Sufficient pro-duction of butyrate by the gut microbiota is considered essential for the maintenance of host health.18 R. callidus affects the intestinal microbial community and human health through the products it creates by break-ing down other complex polysaccharides, including starch and xylan.19 It has been shown that increased B. bifidum dominance promotes the expression of anti-inflammatory cytokines and tight junction proteins in eukaryotic cells.20 B. bifidum has been shown to induce TH1 and TH2 cytokines.21
In this study, we investigated the effect of probiotic use containing B. subtilis on disease severity and inflammatory parameters in patients diagnosed with diarrheal-type IBS. We found that there was a signifi-cant improvement in the IBS-SSS of the patients after the treatment.
The limitations of this study are that it was not a placebo-controlled study, most patients had mild disease severity (mean IBS-SSS < 150), duration of treatment was short, and small bowel bacterial overgrowth syndrome was not tested. Our results can be applied only to relatively healthy population given all of their exclusion criteria.
CONCLUSION
In conclusion, it was demonstrated that probiotic treatment of B. subtilis may increase the dominance of beneficial bacteria such as B. crossotus, R. callidus, and B. bifidum. In patients with diarrheal-type IBS bacil-lus subtilis may reduce the severity of the disease. Studies with larger numbers of patients and with control groups are needed to confirm our findings and investigate the effect of B. subtilis on inflammation.




 Azar Abiyev1
Azar Abiyev1