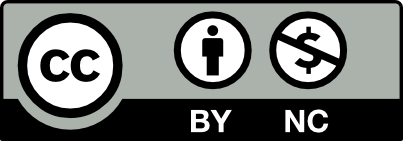
Abstract
Gastric cancer is a heterogeneous disease with the origin of familial and sporadic disease; gastric cancer initiation and progression are caused by various factors such as genetic, epigenetic, and environmental risk factors. Epigenetic alterations determine cell behavior by modulating the transcriptional availability of diverse parts of the genome through DNA methylation, chromatin modification, histone modifications, and non-coding RNAs. DNA methyltransferases catalyze DNA methylation, and epigenetic studies proposed that DNA methylation interacts with histone modifications resulting in a change in gene expression. The multiple crucial biological effects of EZH2 have been identified including signal transduction, DNA repair, cell division, and autophagy. Enhanced EZH2 expression is a biomarker of metastasis in various cancers, such as prostate, breast, hepatocellular carcinomas, colorectal cancer, lung cancer, and gastric cancer. It has been shown that non-coding RNAs, siRNAs, and pharmacological inhibition suppressed the malignant progression of gastric cancer via EZH2 regulation. In this review, we discuss the importance and roles of EZH2 in gastric cancer progression and chemotherapy resistance.
GASTRIC CARCINOMA AND ETIOLOGY
The second leading cause of death among all malignancies and the world’s fourth most frequent cancer is gastric carcinoma. Gastric cancer is a heterogeneous disease with the origin of familial and sporadic disease; gastric cancer initiation and progression are caused by various factors such as genetic, epigenetic, and environmental risk factors.1,2
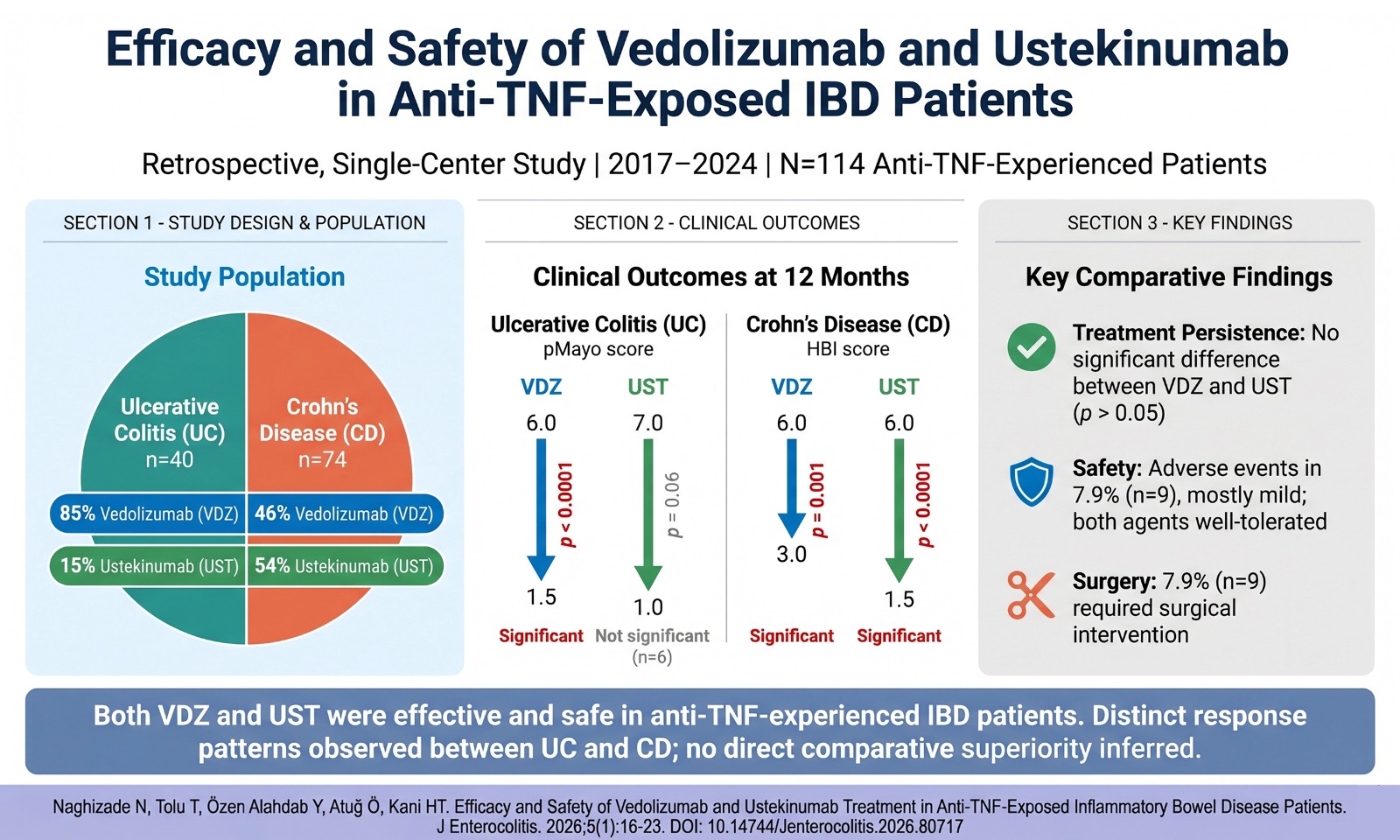
Epigenetic alterations have been described as being correlated with the diagnosis and treatment of gastric cancer patients. More than 90% of all gastric cancers are related to Helicobacter pylori infection, and about 10% of gastric cancers are familial. Sporadic gastric cancer develops in a model of chronic inflammation, intestinal metaplasia, dysplasia, and eventually adenocarcinoma. Sporadic gastric cancer is defined by typical epigenetic modifications but limited genetic alterations across the stages.3 Epigenetic alterations determine cell behavior by modulating the transcriptional availability of diverse parts of the genome through DNA methylation, chromatin modification, histone modifications, and non-coding RNAs (e.g., micro RNAs, long non-coding RNAs, and small interfering RNAs).4 Chromatin remodeling and DNA methylation are 2 crucial epigenetic alterations; chromatin remodeling takes place through histone modifications (principally on the N terminal tails) that finally affect the interplay of DNA and chromatin-modifying proteins, whereas DNA methylation is a chemical alteration in the DNA sequence that frequently occurs in CpG dinucleotides via cytosine methylation.5,6
Helicobacter pylori is a type I carcinogen and an overpowering etiological factor for gastric cancer. H. pylori is well known to stimulate hypermethylation and silencing of various tumor-suppressor genes in H. pyloriinfected non-cancerous and cancerous gastric mucosa. Hypermethylation of tumor-suppressor genes such as CDKN2A, MHL1, MGMT, PTEN, and RUNX3 has been related to the initiation of tumorigenesis and gastric cancer progression.7,8 WW domain-containing oxidoreductase (WWOX) is a tumor-suppressor gene, which was associated with H. pylori, and has been found to be decreased in gastric cancer. Also, H. pylori-infected gastric cancer cell lines demonstrated hypermethylation of WWOX, which was linked with H. pylori-induced increased expression of DNMT1 and DNMT3.9
EPIGENETIC ALTERATION IN GASTRIC CANCER
DNA methyltransferases (DNMTs) catalyze DNA methylation, and epigenetic studies proposed that DNA methylation interacts with histone modifications, resulting in the change in gene expression.10 The expression of enhancer of zeste homolog 2 (EZH2) as a histone methyltransferase has been reported in a few types of cancer. The multiple crucial biological effects of EZH2 have been identified including signal transduction, DNA repair, cell division, and autophagy (Figure 1).11-14
Enhanced EZH2 expression is a biomarker of metastasis in various cancers, such as prostate, breast, hepatocellular carcinomas, colorectal cancer, and lung cancer. Enhancer of zeste homolog 2 histone methyltransferase has been suggested as a new therapeutic target. More recently, overexpression of EZH2 has been revealed in gastric cancer; nevertheless, the clinical importance and molecular mechanisms of EZH2 controlling gastric cancer progression are well-known. Enhancer of zeste homolog 2 is a member of the polycomb repressive complex 2 (PRC2), which catalyzes trimethylation of histone H3 lysine 27 (H3K27me3) and initiates carcinogenesis via epigenetically silencing tumor-suppressor genes.10,14-16
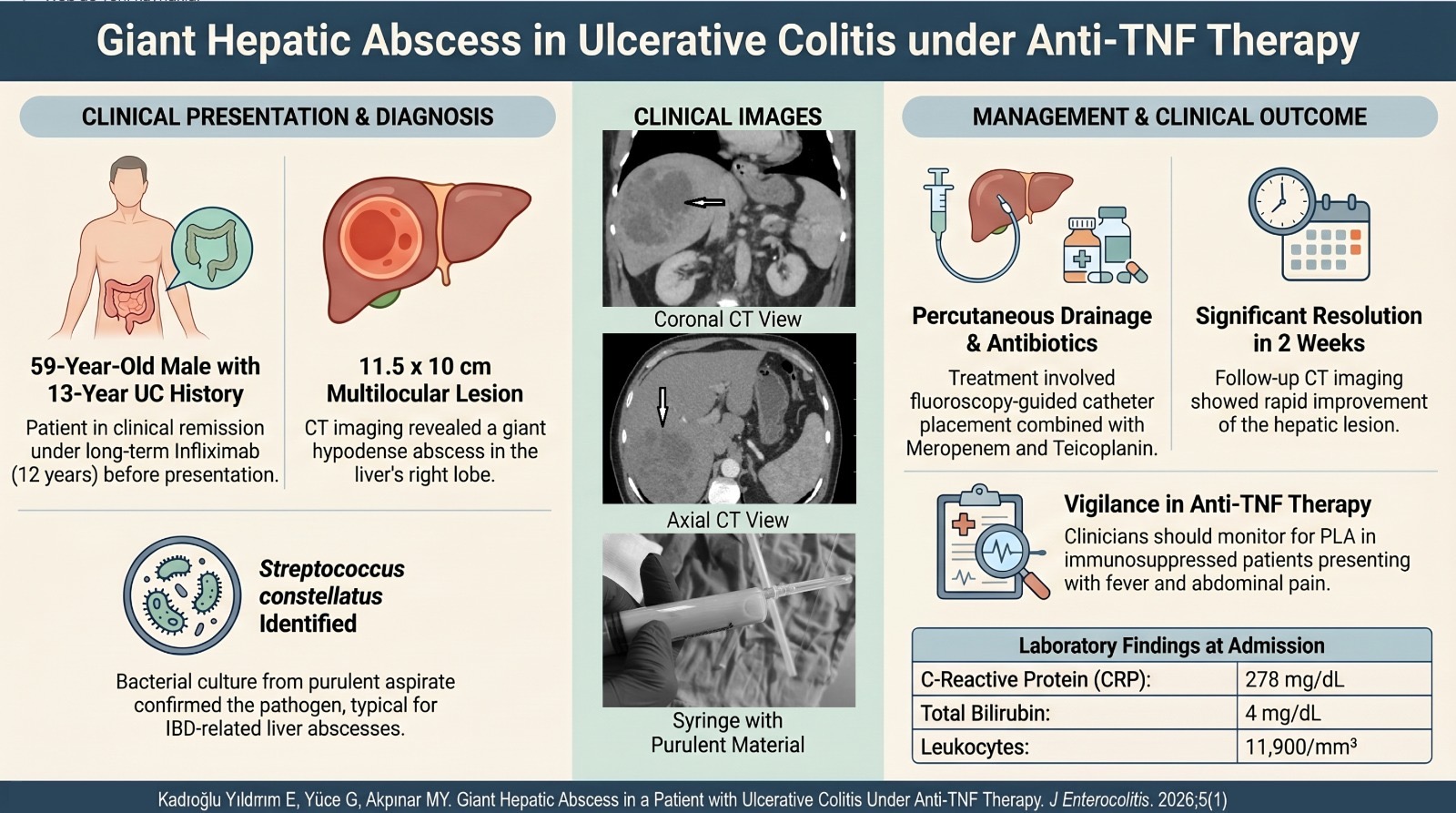
ENHANCER OF ZESTE HOMOLOG 2 AND SIGNALING PATHWAYS INVOLVED IN GASTRIC CANCER
Bone morphogenetic proteins (BMPs) are a member of transforming growth factor-beta and function through the BMP-SMAD (Suppressor of Mothers against Decapentaplegic) pathway. For the first time, BMPs were found to be proteins that stimulate ectopic bone generation and initiate the development of gastrointestinal organs. Bone morphogenetic proteins have crucial roles in cell proliferation and apoptosis in numerous cancers such as gastric cancer. Many studies have shown that BMP2 modulates the expression of EZH2. However, as a tumor-suppressor gene, BMP2 might suppress the EZH2 expression and H3K27me3 through a new SMAD-BMP pathway, leading to a reduc-tion in CDK2, CDK4, and CDK6 and enhancement in P21 and P16.17
Phosphatase and tensin homolog (PTEN) is a straight target of EZH2. Most significantly, EZH2 can bind to the PTEN locus, decrease PTEN expression, and activate the Akt pathway. Besides, the EZH2–PTEN axis upregulates vimentin and decreases E-cadherin, thus finally leading to the acquisition of the epith elial –mese nchym al transition in gastric cancer cells.18
Inflammation plays a significant role in gastric cancer progression, and anomalous IL-6/STAT3 signaling pathways in cancer cells have been identified as an important mechanism for the onset and development of cancer. Interleukin-6 stimulates the signal transducer and activator of transcription 3 (STAT3), resulting in epigenetic abnormality during tumorigenesis, especially in the expression of the EZH2 histone meth-yltransferase enzyme. EZH2 binds to STAT3 and methylates it, causing increased STAT3 activity by enhancing STAT3 tyrosine phosphoryla-tion. An EZH2 inhibitor reverses the inhibiting of polycomb target genes and reduces STAT3 activity. Consequently, targeting STAT3, p-STAT3, and EZH2 might be a sensitive medicine and new therapy for gastric cancer patients.18,19
NON-CODING RNAS AND ENHANCER OF ZESTE HOMOLOG 2 IN GASTRIC CANCER
MicroRNAs (miRNAs) are small RNAs (about 22 nt) that have a sig-nificant role in modulating gene expression in tumor samples and are interesting as helpful biomarkers for early phase prognosis and diagnosis of cancer. Various microRNAs have been linked with gastric cancer and its prognosis. In cohort studies, miR-221, miR-376c, and miR-744 were defined as being talented in distinguishing gastric cancer patients.20,21
Chromobox protein homolog 7 (CBX7), an element of the polycomb group family, is linked to the regulation of cancer cell proliferation, progression, and metastasis. The CBX7 overexpression is reported in gastric tumors. AKT-NF-κB-miR-21 signaling axis has been indicated as a critical effector of CBX7 in gastric cancer stem cell initiation and expansion.21 The studies showed that miR-625-3p, miR-217, and miR-137 were closely correlated with lymph node metastasis, remote metas-tasis, and poor prognosis in gastric cancer patients. They also showed that miR-625-3p, miR-217, and miR-137 suppressed the malignant progression of gastric cancer via EZH2 regulation.22-24
Long non-coding RNAs (lncRNAs) are non-coding RNAs that have no protein-coding potential and are longer than 200 nt. Long non-coding RNAs have been identified as critical regulators of biological activities and various cancers through their interplay with multiple biomolecules. For example, C8orf76 as an oncogene directly targets lncRNA-DUSP5P1 for its transcriptional stimulation, consequently activating the MAPK/ERK signaling pathway and initiating gastric tumorigenesis. Recent studies have investigated Linc01503 in gastric cancer and have indicated that it is closely associated with the overall survival of gastric cancer patients. Linc01503 could promote gastric cancer cell invasion via activating the wnt signaling pathway and interacting with early growth response protein 1 (EGR1). Furthermore, the knockdown of linc01503 resulted in the induction of apoptosis and the G1/G0 phase stopping in gastric cancer. Also, these studies reported that the interaction of EZH2 with lysine (K)-specific demethylase 1A (LSD1) resulted in the silencing of cyclindependent kinase inhibitor 1A (CDKN1A) and dual-specificity phosphatase 5 (DUSP5) and mediated cell cycle progression and tumorigenesis in gastric cancer. Consequentially, the EGR1/ linc0 1503/ EZH2/ DUSP5 /CDKN 1A axis may be an appealing target for gastric cancer therapeutics.25,26
Another study showed that LINC00337 as an oncogene was increased in the gastric cancer cells, and the overexpression of LINC00337 defines the poor clinical outcome. The in vitro and in vivo studies showed that reducing LINC00337 suppressed invasion, proliferation, and tumorigenesis. It was also reported that LINC00337 epigenetically inhibited p21 through EZH2-mediated blocking.27 Small nucleolar RNA host gene 6 (SNHG6) has an important role in the progression of gastric cancers. Knockdown of lncRNA SNHG6 suppresses gastric cancer development through overexpression of p21. Also, the inhibition of SNHG6 and the increase of p21 have resulted in the activation of the JNK pathway and repression of EZH2. Thus, SNHG6 plays a significant role in gastric cancer progression.28
A new study shows that LINC00460 as an independent prognostic marker is upregulated in gastric cancer tissues compared to normal tissues. Investigation of the function of LINC00460 has shown that LINC00460 initiated gastric cancer proliferation via the decrease in cyclin G2, which is modulated by LSD1 and EZH2 proteins.29
ENHANCER OF ZESTE HOMOLOG 2 INHIBITORS IN GASTRIC CANCER
More studies have shown the role of EZH2 in cancer pathophysiology. RNA-based silencing such as siRNAs and shRNAs have been widely utilized to inhibit and modulate EZH2 signaling in cancer experimental research. Pharmacological and biological inhibition of EZH2 has been an area for enhancement of research efforts. Enhancer of zeste homolog 2 inhibitors include GSK926, GSK343, 3-deazaneplanocin A (DZNep), valemetostat, and tazemetostat (E7438/EPZ6438).
The strategies to suppress EZH2 contain inhibition of double EZH1/EZH2 and inhibitors that disrupt the PRC2 structure. Other strategies for the degradation of EZH2 are attempts to deliver specific drugs and siRNAs together with nanoparticles.30-32 Cisplatin and oxaliplatin are a member of platinum drugs and the first-line chemotherapy for gastric cancer. Cisplatin resistance is an important strategy in gastric cancer chemotherapy. The inhibition of EZH2 by siRNA improves cisplatin resistance in AGS gastric cancer cells.33 Another study showed that that miR-625-3p, miR-217, and miR-137 suppressed the malignant progression of gastric cancer via EZH2 regulation.22-24
Long non-coding RNAs (lncRNAs) are non-coding RNAs that have no protein-coding potential and are longer than 200 nt. Long non-coding RNAs have been identified as critical regulators of biological activities and various cancers through their interplay with multiple biomolecules. For example, C8orf76 as an oncogene directly targets lncRNA-DUSP5P1 for its transcriptional stimulation, consequently activating the MAPK/ERK signaling pathway and initiating gastric tumorigenesis. Recent studies have investigated Linc01503 in gastric cancer and have indicated that it is closely associated with the overall survival of gastric cancer patients. Linc01503 could promote gastric cancer cell invasion via activating the wnt signaling pathway and interacting with early growth response protein 1 (EGR1). Furthermore, the knockdown of linc01503 resulted in the induction of apoptosis and the G1/G0 phase stopping in gastric cancer. Also, these studies reported that the interaction of EZH2 with lysine (K)-specific demethylase 1A (LSD1) resulted in the silencing of cyclin-dependent kinase inhibitor 1A (CDKN1A) and dual-specificity phosphatase 5 (DUSP5) and mediated cell cycle progression and tumorigenesis in gastric cancer. Consequentially, the EGR1/ linc0 1503/ EZH2/ DUSP5 /CDKN 1A axis may be an appealing target for gastric cancer therapeutics.25,26
Another study showed that LINC00337 as an oncogene was increased in the gastric cancer cells, and the overexpression of LINC00337 defines the poor clinical outcome. The in vitro and in vivo studies showed that reducing LINC00337 suppressed invasion, proliferation, and tumorigenesis. It was also reported that LINC00337 epigenetically inhibited p21 through EZH2-mediated blocking.27 Small nucleolar RNA host gene 6 (SNHG6) has an important role in the progression of gastric cancers. Knockdown of lncRNA SNHG6 suppresses gastric cancer development through overexpression of p21. Also, the inhibition of SNHG6 and the increase of p21 have resulted in the activation of the JNK pathway and repression of EZH2. Thus, SNHG6 plays a significant role in gastric cancer progression.28
A new study shows that LINC00460 as an independent prognostic marker is upregulated in gastric cancer tissues compared to normal tissues. Investigation of the function of LINC00460 has shown that LINC00460 initiated gastric cancer proliferation via the decrease in cyclin G2, which is modulated by LSD1 and EZH2 proteins.29
ENHANCER OF ZESTE HOMOLOG 2 INHIBITORS IN GASTRIC CANCER
More studies have shown the role of EZH2 in cancer pathophysiology. RNA-based silencing such as siRNAs and shRNAs have been widely utilized to inhibit and modulate EZH2 signaling in cancer experimental research. Pharmacological and biological inhibition of EZH2 has been an area for enhancement of research efforts. Enhancer of zeste homolog 2 inhibitors include GSK926, GSK343, 3-deazaneplanocin A (DZNep), valemetostat, and tazemetostat (E7438/EPZ6438).
The strategies to suppress EZH2 contain inhibition of double EZH1/EZH2 and inhibitors that disrupt the PRC2 structure. Other strategies for the degradation of EZH2 are attempts to deliver specific drugs and siRNAs together with nanoparticles.30-32 Cisplatin and oxaliplatin are a member of platinum drugs and the first-line chemotherapy for gastric cancer. Cisplatin resistance is an important strategy in gastric cancer chemotherapy. The inhibition of EZH2 by siRNA improves cisplatin resistance in AGS gastric cancer cells.33 Another study showed that DZNep as a histone methyltransferase inhibitor reverses cisplatin resis-tance of gastric cancer cells through the inhibition of HIF-1α, Wnt/β-catenin signaling molecules, and EZH2 protein.34 Nevertheless, the knockdown of lncRNA HOXD cluster antisense RNA 1 (HOXD-AS1) has reversed cisplatin resistance through the epigenetic silencing of programmed cell death 4 via binding to EZH2 in gastric cancer cells.35
CONCLUSION
Enhanced EZH2 expression is a biomarker of metastasis in various cancers, such as prostate, breast, hepatocellular carcinomas, colorectal cancer, lung cancer, and gastric cancer. Nevertheless, the clinical importance and molecular mechanisms of EZH2 controlling gastric cancer progression are well known. This review provided roles of the dysregulation of EZH2 in gastric cancer and possible mechanisms for the suppression of EZH2 activity.




 Nalan Gülşen Ünal1
Nalan Gülşen Ünal1